Read the full article on Science Direct
The explosive spread of Zika virus (ZIKV) and its linkage to neurodevelopmental disease in infants born to women infected while pregnant presents a new global health threat and highlights the need for a vaccine. Understanding features of the virion recognized by neutralizing antibodies will help inform vaccine development. ZIKV particles are coated with envelope (E) proteins, which mediate host entry and are targeted by neutralizing antibodies. To better understand determinants of antibody recognition, we replaced ZIKV E residues that form a loop including a single N-linked glycan with the analogous residues of the related dengue virus (DENV). We observed a large increase in ZIKV sensitivity to neutralization by antibodies targeting epitopes predicted to be poorly exposed on wildtype virions, but only minimal effects on the neutralization potency of antibodies targeting highly accessible epitopes. We propose that the ZIKV E glycan loop modulates antibody recognition by altering E protein conformational flexibility, which allows the transient exposure of otherwise cryptic targets.
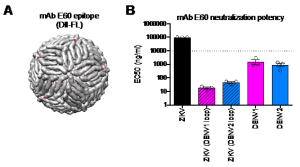
(A) The location of the poorly exposed DII-FL epitope targeted by monoclonal antibody (mAb) E60 is indicated by red dots on the E proteins coating the surface of the mature ZIKV particle. (B) Average mAb E60 concentrations required to inhibit virus infectivity by 50% (EC50) were obtained from three independent experiments with ZIKV, DENV1, DENV2, and ZIKV with the E protein glycan loop replaced with that of DENV1 [ZIKV (DENV1 loop)] or DENV2 [ZIKV (DENV2 loop)]. The dotted horizontal line represents the highest antibody concentration tested. At this concentration, 50% inhibition was not achieved for ZIKV.
Our work was motivated by the near-simultaneous determination of a high-resolution structure of the mature form of the ZIKV virion by research teams at Purdue and Duke-NUS. These new structures revealed ZIKV to be very similar to the closely related DENV. However, both groups noted that the largest difference between these viruses was the E protein loop to which an N-linked sugar is attached. By comparison to DENV, the E protein glycan loop of ZIKV extends further towards the highly conserved fusion loop (DII-FL) on the neighboring E protein within the antiparallel dimer on the virion surface; the sugar at this position had been suggested to stabilize this dimer. ZIKV is largely insensitive to neutralization by cross-reactive antibodies targeting the DII-FL epitope. Although this epitope is poorly accessible for antibody recognition on mature virions, it may become transiently exposed by conformational dynamics or ‘breathing.’ We hypothesized that the extended ZIKV glycan loop could modulate antibody recognition directly via local steric effects, or indirectly via overall changes in E protein breathing. Our findings support the latter hypothesis, as ZIKV containing the DENV glycan loop displayed increased sensitivity to neutralization by antibodies targeting not only the proximal DII-FL, but also distal cryptic epitopes.
We were surprised by a number of our findings. First, although sugars on viral glycoproteins have been shown to impact antibody recognition, the increased sensitivity of ZIKV chimeras containing the DENV glycan loop residues to neutralization was independent of the presence of a sugar attached to this loop. Second, reciprocal DENV chimeras containing the ZIKV glycan loop similarly displayed increased sensitivity to neutralization by E-FL-specific antibodies, suggesting that limited accessibility of ZIKV E-FL is unlikely due to local steric effects involving a larger glycan loop. Altogether, these data provide another example of how changes in the E protein alter virion antigenic structure in unexpected ways.
About the Authors

Leslie Goo, Group Leader, Infectious Disease Initiative, Chan Zuckerberg Biohub, San Francisco, California, United States of America.
About the Research
The Zika virus envelope protein glycan loop regulates virion antigenicity
Virology, Volume 515, February 2018, Pages 191-202
