Read the full article on ScienceDirect
The T7 ejection nanomachine components gp15–gp16 form a spiral ring complex that binds DNA and a lipid membrane
Text by Sebastian Leptihn and Andreas Kuhn
Most people may have heard of T7 phage only from its promoter found in many plasmids used for high level expression in strains carrying the T7 RNA polymerase gene. However, there is so much more to this small virus, that belongs to the family of Podoviridae with the icosahedral shape and a short, stubby tail.
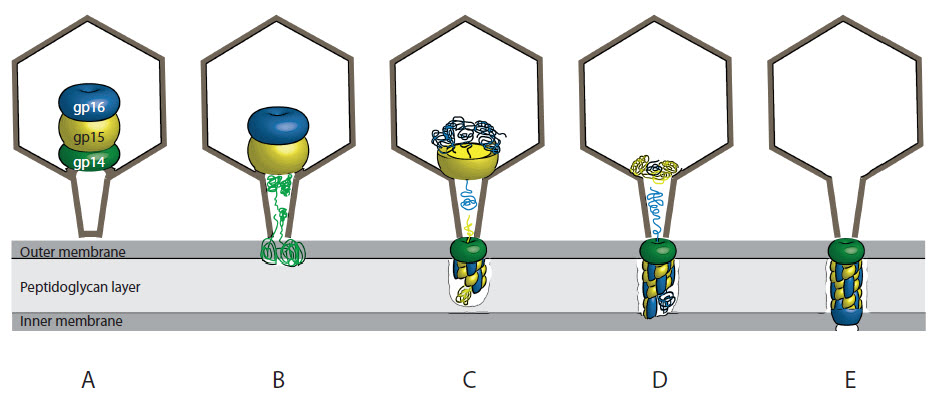
This work started decades ago when the senior author Andreas Kuhn was a young PhD student and some authors were still crawling around in diapers. In Rudi Hausmann’s and Ed Kellenberger’s labs, Andreas discovered the importance of the T7 phage gene 16. Without it, the phage lost its ability to assemble progeny in the host cells and aberrant empty capsids appeared inside the cells visualized by electron microscopy. It is fascinating, that in the center of the capsid, the phage carries syringe components to inject DNA into the host. It doesn’t have them pre-assembled ready for injection by simply extending a drill into the host cell; no, it carries a nicely packed parcel of the drill proteins tightly packed in a structure called the “inner core”. It is like a parcel from IKEA that -with the proper manual- allows you to build a piece of furniture. The phage has no manual, but still, a complex called the ejectosome is assembled in the host cell wall. This was one of our major questions: How does the phage assemble the DNA channel?
Back to the analogy: If the phage was a van, delivering the parcel containing the ejectosome, without opening the loading ramp, it manages to deliver the parcel through a small window that is smaller than the parcel itself. The window in the phage is the “portal complex” that creates an opening too small to allow a folded protein to slip through. How then do the ejectosome proteins translocate? The “inner core complex” needs to disintegrate, the proteins need to unfold at least partially and then refold and assemble again. Amazingly, the proteins form a completely different structure outside that phage head, the ejectosome.
While working on this exciting project, Domenico had observed that two of these proteins spontaneously form a 2:1 complex. Sebastian looked into the folding of each of these proteins and was stunned by their refolding capacities, explaining how the proteins are able to exit the phage head and form the ejectosome. Max checked the structure of the complex by electron microscopy and we were amazed to see spiral filaments that looked very different from the structures these proteins appear inside the phage capsid. Gisela found that the complex of the two proteins binds to protein-free liposomes and DNA, explaining how they might bind the host membrane and allow the transport of the phage genome into the host cell.