Read the full article on ScienceDirect
The main reason that HIV is difficult to cure is because it can hide from the immune system and anti-HIV drugs by entering a latent “sleeping” state within its host cells. One approach to eliminate this latent virus is to “wake it up” and cause it to express viral proteins, allowing the host cell to be killed by the virus or by the immune response to the expressed viral proteins. We had previously shown that certain compounds inspired by natural molecules but designed and made in a lab were more effective than the natural agents at waking up latent HIV and causing the infected host cells to be killed (Marsden et al., 2017). In the current study we expanded this work to show that these synthetically tunable compounds also exert a variety of potentially useful effects on immune cells, including reducing levels of HIV entry receptor expression, inhibiting HIV spread in culture, and working even better when used in combination with other classes of compounds (synergizing).
This study is part of a longstanding (over 10-year) collaboration between the Wender bioorganic chemistry laboratory at Stanford, and the Marsden and Zack virology labs at UCLA. The ideas from this study were derived in part from previously published observations regarding how natural PKC modulators affect cell receptor levels and synergize with a class of latency reversing agents known as histone deacetylase inhibitors (HDACi). However, it was unknown whether this is a general feature of PKC modulators, and (importantly) whether synthetic PKC modulators that have been designed and synthesized in a lab would have similar effects. In addition to the previously documented potent latency reversal and better in vivo tolerability that these compounds exhibit, we soon realized that their ability to wake up latent HIV could be amplified by combination with HDACi and that these new molecules did indeed inhibit HIV spread (Fig 1). One surprise along the way was when we quantified proinflammatory cytokine production in primary cells exposed to these compounds and found substantial variability in proinflammatory cytokine production in cells treated with different PKC modulators, suggesting that specific structural variations can differentially influence downstream cytokine production.
Together this new work suggests that the synthetic molecules we are exploring can not only wake up the latent HIV but also help prevent any virus that is produced from infecting new cells and causing more damage to the host. It also points the way to combinations of drugs that might work better than either drug alone.
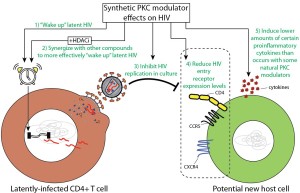
Figure 1. We identified several characteristics of synthetic PKC modulators that may prove important in HIV cure approaches (green text). The parameters examined were guided in part by previously published observations with natural PKC modulators.
Reference
Marsden, M.D., Loy, B.A., Wu, X., Ramirez, C.M., Schrier, A.J., Murray, D., Shimizu, A., Ryckbosch, S.M., Near, K.E., Chun, T.W., Wender, P.A., Zack, J.A., 2017. In vivo activation of latent HIV with a synthetic bryostatin analog effects both latent cell “kick” and “kill” in strategy for virus eradication. PLoS pathogens 13, e1006575.
Introducing the authors

Matthew D. Marsden, Ph.D. (left), Department of Medicine, Division of Hematology and Oncology, University of California Los Angeles, Los Angeles, United States
Paul A. Wender, Ph.D. (middle), Departments of Chemistry and of Chemical and Systems Biology, Stanford University, Stanford, United States
Jerome A. Zack, Ph.D. (right), Department of Medicine, Division of Hematology and Oncology, University of California Los Angeles, Los Angeles,United States
About the research
Characterization of designed, synthetically accessible bryostatin analog HIV latency reversing agents
Virology, Volume 520, July 2018, Pages 83-93
