Read the full article on ScienceDirect.
An in vitro reprogrammable antiviral RISC with size-preferential ribonuclease activity
Text by Herman Scholthof
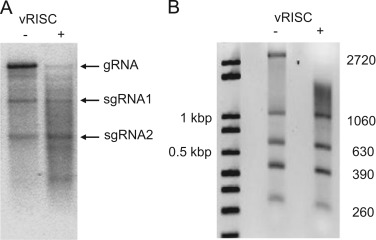
It has been recognized now for about 15 years that plants, like other eukaryotes, have evolved a defense mechanism to target and destroy virus RNA to combat the infection. The components of the silencing pathway are mostly genetically identified but biochemical properties of one of the most vital units, the RNA-induced silencing complex (RISC) that actually finds and cleaves the viral RNA, remain somewhat elusive. We have isolated the virus-activated RISC from infected plants and showed that it is an active sequence-specific endonuclease in vitro, and this activity depends on the siRNA that is used to program RISC. An unexpected finding was that the RISC was preferentially targeting longer RNAs of more than 1 kb. Although the biochemical mechanism for this property remains unknown, it agrees with published findings by us and others that very short virus-derived defective RNAs of ~0.5 kb are not sensitive to the silencing defense in infected plants.
The idea for this study came from experimental results while analyzing the nuclease properties of virus-activated RISCs isolated from plants infected with different viruses. We had previously shown that an active RISC could be isolated from infected plants and the main purpose or objective was then to demonstrate that the nuclease activity was sequence-specific, as would be expected for RISC. However, upon close examination of results obtained from experiments conducted for different reasons, more and more the data seemed to suggest that no matter the objective of the study, in addition to sequence-specificity, virus RNAs of different lengths seemed to vary in their susceptibility to RISC-mediated degradation. We were intrigued by this observation and in the present study we provide evidence that this is a consistent property during our in vitro conducted experiments. The ‘aha’ moment was when we realized that it was well documented that in infected plants short virus RNAs are somehow protected from RISC. Further studies are needed but we are excited about the possibility that our developed in vitro system may lead to a thorough understanding of the underlying biochemical principle. In the larger scheme of things we think it is possible that our finding hints at the possibility that a virus-activated RISC that prefers to target longer RNAs may contribute to providing a safety mechanism to avoid too many off target effects on host mRNAs.